 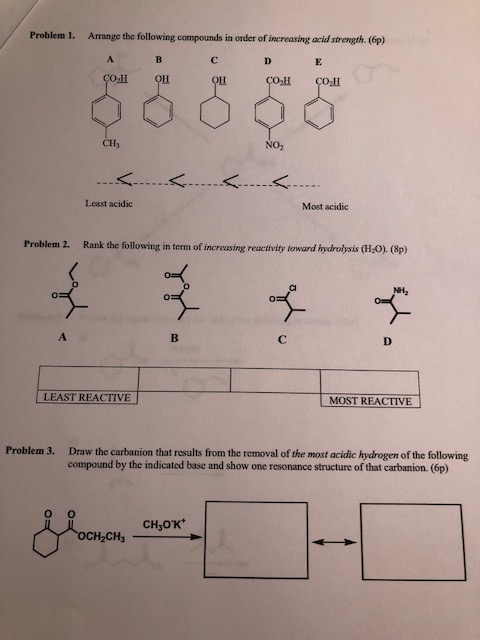
Write the structures of A, B, C, D, and E in the following reactions: of iodoform on heating with NaOH and l 2, it is CH 3COCH 3.ĬH 3COCH 3 + 3I 2 + 4NaOH → CHI 3 + CH 3COONa + 3Nal + 3H 2Oī does not give iodoform, it is CH 3CH 2CHO. (v) Two isomers are CH 3COCH 3 and CH 3CH 2CHO (iv) CH 3 CH 2 CH = CH CH 2 CN CH 3 CH 2 CH = CH CH 2 CHO Therefore, pKa of ClCH 2COOH is less than that of CH 3COOH. Consequently, Cl - CH 2COOH is more acidic than acetic acid. As a result, the electrons are withdrawn more strongly towards 0 of the O-H bond and promote the release of the proton. It withdraws the electrons from the C to which it is attached and this effect is transmitted throughout the chain. (iii) Chlorine is an electron-withdrawing group (-I inductive effect). (b) Add ammoniacal AgNO 3 solution (Tollen’s reagent), HCOOH gives silver mirror while CH 3CH 2COOH does not. C 6H 5CH = CHCOCH 2CH 3 does not give yellow ppt. (ii) (a) Heat both the compounds with NaOH and l 2, C 6H 5 CH = CHCOCH 3 gives yellow ppt. On heating with NaOH and l 2, isomer A forms a yellow precipitate of iodoform whereas isomer B does not form any precipitate. (v) A and B are two functional isomers of compound C 3H 6O. (iv) Write the product in the following reaction. 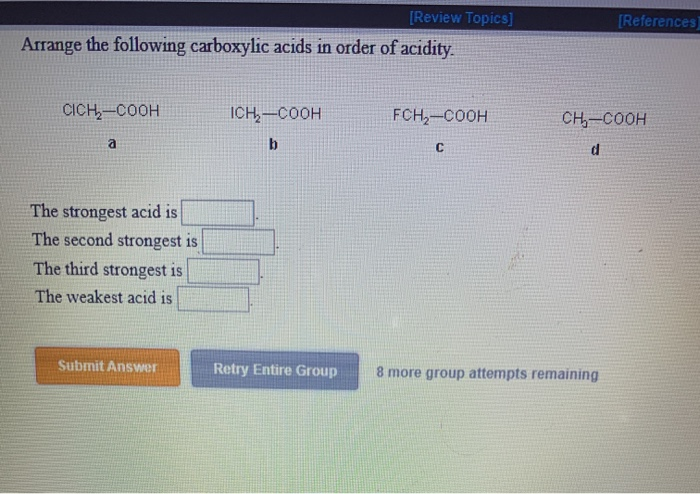
(iii) Why pKa of Cl-CH 2-COOH is lower than the pK a of CH 3COOH? (ii) Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction: (i) Write the chemical reaction involved in the Etard reaction. (iii) Arrange the following in the increasing order of their boiling points: (a) C 6H 5 – CH = CH – COCH 3 and C 6H 5 – CH = CH – CO CH 2CH 3 (i) Write the structures of A, B, C, and D in the following reactions: (iii) CH 3CH 2CH 2COCH 3 gives iodoform test The poisoning of palladium catalyst decreases its activity and it does not allow the further reduction of an aldehyde into alcohol. The reaction is carried out by passing through a hot solution of the acid chloride in the presence of palladium deposited over barium sulfate (partially poisoned with sulfur or quinoline). Acid chlorides are converted to corresponding aldehydes by catalytic reduction. They give the characteristic reactions of the group. But no resonance is possible in aldehydes and ketones. In carboxylic acids, the carbonyl group is involved in resonance, as follows: (CBSE 2012)īutanone C = 0) as given by aldehydes and ketones. (CBSE 2011, CBSE Delhi 2013)Īrrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions:Įthanal, propanal, propanone, butanone. (CBSE 2010)ĭraw the structure of 3-methylbutanal. (CBSE Delhi 2008, CBSE AI 2011, 2013, 2014)ĭraw the structural formula of a 1-phenyl propane-2-one molecule. (CBSE Delhi 2013, 2014)ĭraw the structure of the compound whose IUPAC name is 4-chloropentan 2-one. Write the structure of the p-Methylbenzal- dehyde molecule. 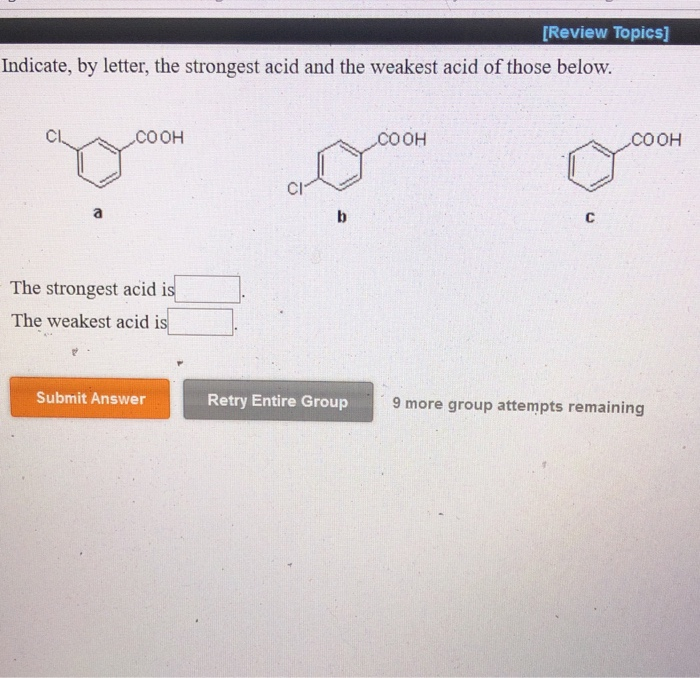
Write the IUPAC name of the following: (CBSE AI 2012) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
